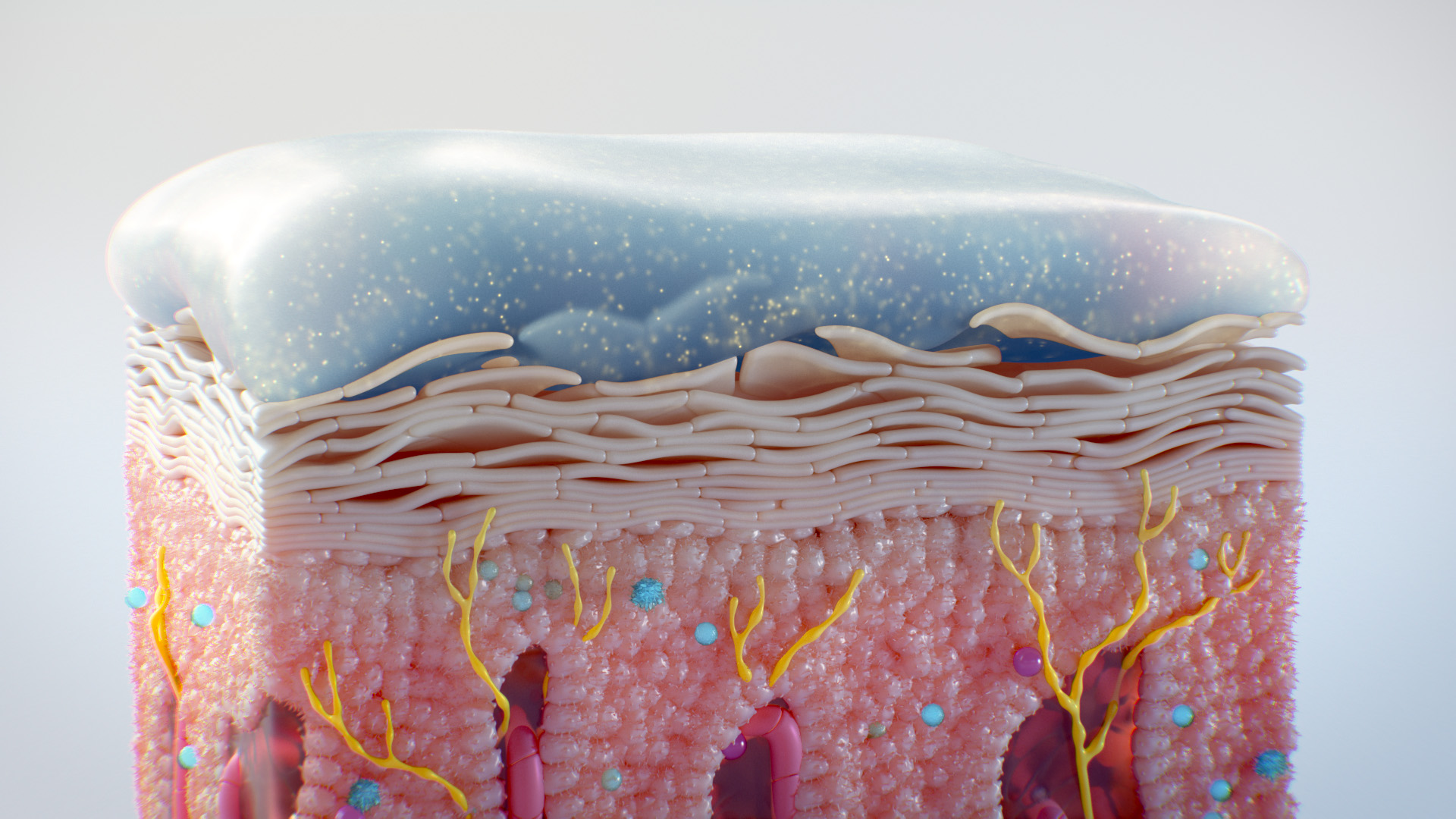
Interactive Medical and Scientific Experiences
Interactive medical and scientific experiences make your messaging easier to understand and impossible to ignore. AXS Studio designs custom apps, websites and conference experiences to help you make more meaningful connections with HCPs. Give your MSLs an edge with dynamic 3D MOD and MOA apps. Our medical device apps function on mobile devices and large touchscreens—perfect for reps in the field and self-guided product demos at conferences. Here are just some of the possibilities: educational games; MOD simulators; experience walls; interactive holograms; migraine photo booths; virtual reality (VR); augmented reality (AR); and mixed reality (MR).

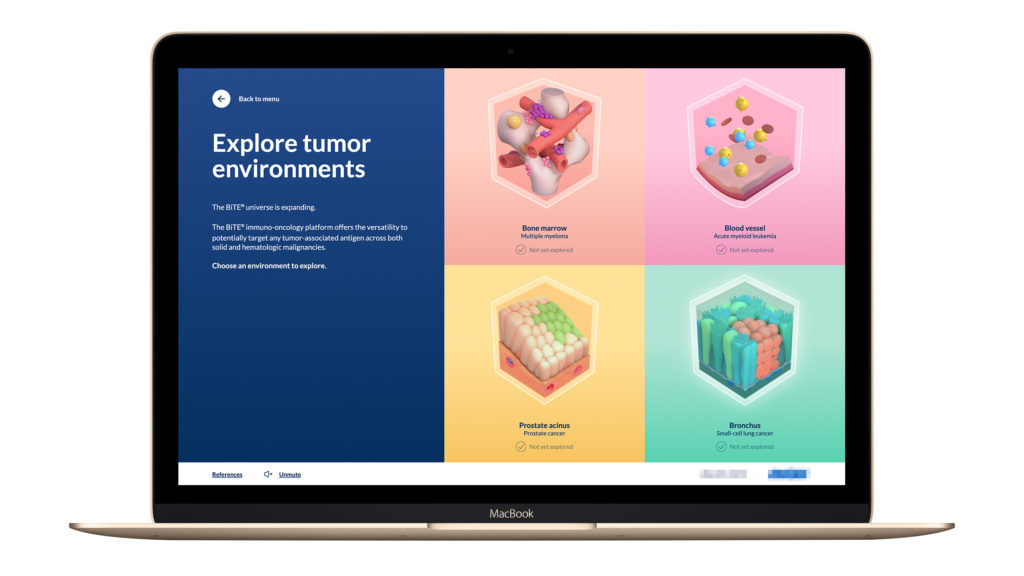
Interactive MOA for Booth and Browser
3D Interactive Experience for Booth and Browser

iPad App: Medical Device Simulator
Eyenovia
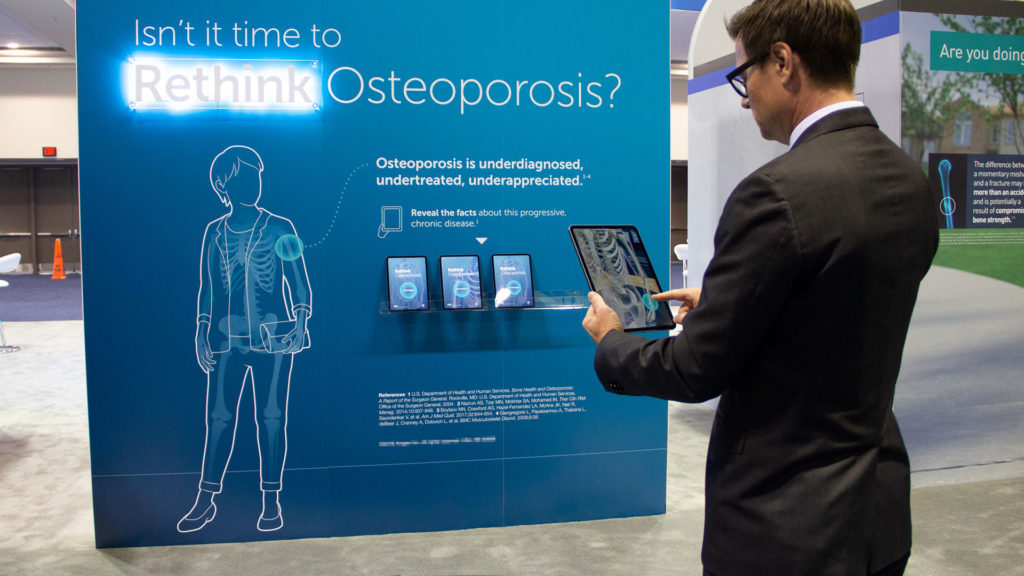
Medical AR: Interactive Osteoporosis Education
Biopharmaceutical Client

Interactive Migraine VR Experience
Biopharmaceutical Client
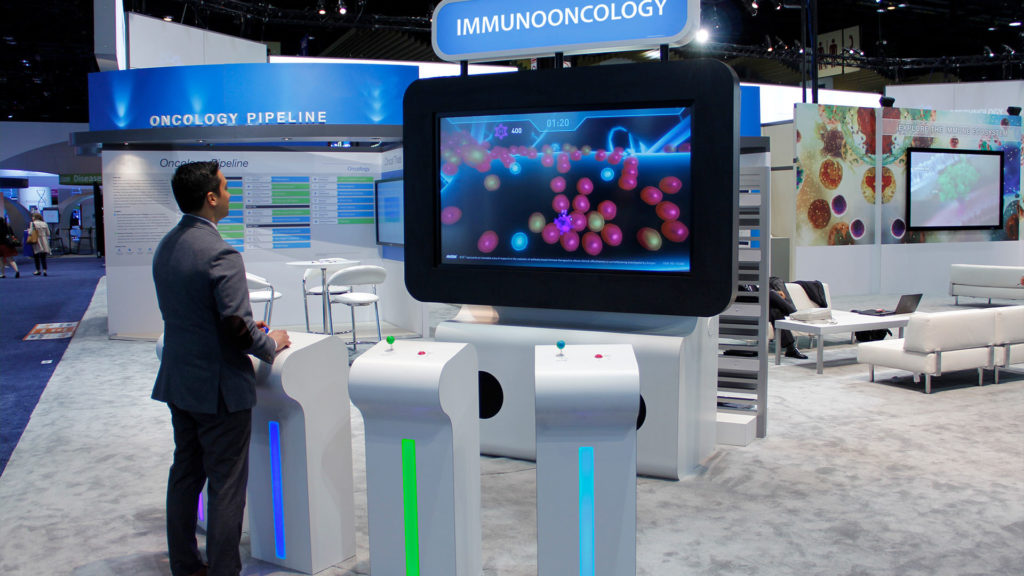
Medical Game: Oncology Arcade Experience
Biopharmaceutical Client

Biosimilar Development Simulator
Biopharmaceutical Client

Medical Mixed Reality (MR): Cardiorenal Disease
Pharmaceutical Client

Multiple Myeloma Conference Exhibit
Biopharmaceutical Client

CVD Explorer interactive Hologram
Biopharmaceutical Client

Medical VR: Cancer Immunotherapy Simulator
Biopharmaceutical Client
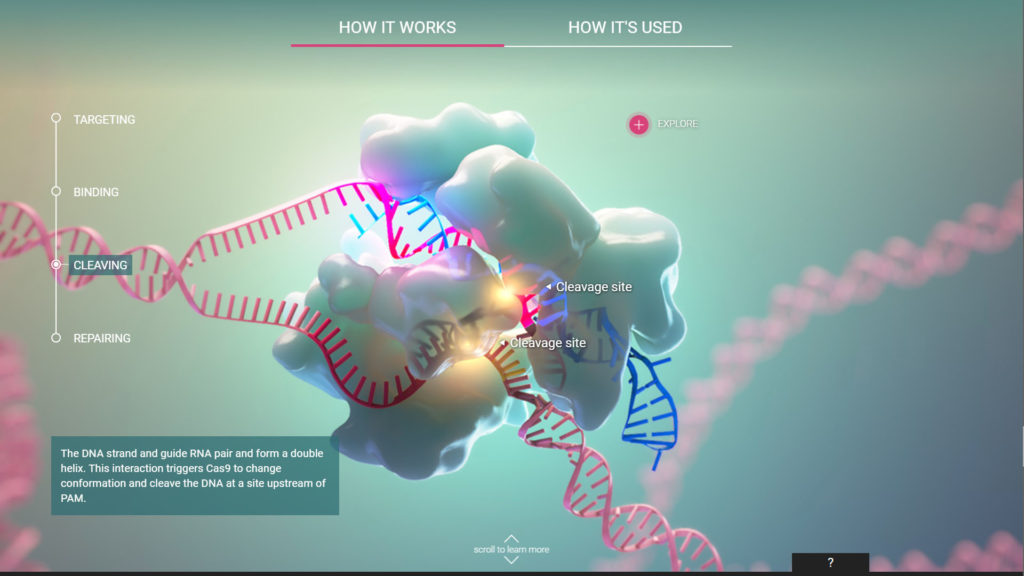
CRISPR Cas-9 Interactive Experience
HHMI Biointeractive
interactive-medical-scientific-experiences
https://axs3d.com/wp-admin/admin-ajax.php
2

It’s been an absolute pleasure working with AXS. They overhauled our scientific materials, providing a consistent look across all our tangible and digital assets to convey our scientific pipeline in a graphically appealing way.
Heather Douangpanya
Director, Global Scientific Communications, Seagen Inc.

Everything from the animation and the stills visually show the story of FaraPulse and the market leading product we set out to achieve. The final deliverables met and even exceeded the original vision. KUDOS and THANKS to you and the team on an AMAZING end product.
Dan Chase
Principal Product Manager, Boston Scientific

We chose AXS Studio because of their unique ability to curate a better, more visually impactful story resonating with both a scientific and non-scientific target audience.
Glen Tinevez
Vice President, Marketing North America Commercial Operations,
Ortho Clinical Diagnostics
Ortho Clinical Diagnostics

The AXS team was a pleasure to work with and I greatly appreciated their creative and scientific expertise. The videos received outstanding feedback and are one of the most popular assets for our disease state education campaign.
Rachel Raynes
Global Medical Communications, Senior Manager | Heart Failure GCCL, AMGEN

What a privilege and blessing it has been working with the AXS team these last few months. The incredible quality and attention to detail in your work, the care you put into navigating relationships, and the commitment you have to pulling off the impossible — it’s been a sight to behold. We are so grateful and appreciative of all your efforts. Thank you!
Susan Bartlett
Principal, Workomics

You guys are doing great work! The animation with the electric field displayed has been an 'AH-HA' moment for all of our Doctors, strategic collaborators and many of our employees!
Allan Zingeler
President & CEO, Farapulse

My previous vendor struggled to conceptually understand the purpose of the project. AXS understood and delivered an amazing product—better than what I initially imagined. We have experienced more engagement with key customers and investors, and the interactive will be a tool we can use across multiple platforms.
Julie Whitcomb
Head of Medical Affairs, Eyenovia

Wow! Just wow! 🤯 mind blown! This is amazing!! My heartfelt gratitude to your team. Their dedication to our project is only outweighed by their creative talents.
John West
Chief Development Officer, SciMar Ltd.

AXS was a true partner in creating highly compelling MOA videos for an important pharma client. They collaborated with us through numerous challenges on a tight schedule and hit our deadline with a product our client loved!
Senior Account Executive, Goodfuse

Thank you for being so great to work with and for creating such an amazing animation. Everyone loves it. Our Director of Sales called it “a game changer”. Our CEO presented the animation in recent talks and shared it with retina specialists. It is a hit! AXS has been a valuable partner and we look forward to working with you again in the future.
Allison Dabney
Director, Marketing, LumiThera

I can always rely on AXS to bring me unique, creative ideas for interactive media that boost traffic and HCP engagement at our congress booths.
Peter Baumeister
Senior Manager, Global Scientific Communications,
Amgen
Amgen

It’s been wonderful working with AXS Studio. The experience has been fantastic, fun, and rewarding. We have had glowing feedback on the video and are finalizing plans to integrate into discussions with all prospective research candidates. Our patient character is crushing it in the Alzheimer’s LP world.
Danielle Goldfarb
MD, Clinical Assistant Professor of Neurology and Psychiatry, Banner Sun Health Research Institute

These are absolutely fantastic visualizations. They convey the real science. I still am incredibly proud of what we did together on this video and I use it in my lectures to Harvard students and externally.
Donald E. Ingber
MD, PhD, Director, Wyss Institute for Biologically Inspired Engineering, Harvard University

The team at AXS are true pros. They asked all of the right questions from the start, from technical/scientific details to overall look and feel, and completed this amazing cover rather quickly as a result.
Kelly Krause
Creative Director, Nature

It is an absolute pleasure to work with the AXS team. Their work is consistently well-researched, beautifully crafted, and press-ready.
Jen Christiansen
Art Director of Information Graphics, Scientific American Magazine

Collaborating with the AXS team on high-impact science visualization projects is always a pleasure. I can rest assured that the final product will be clear, elegant, and beautiful while meeting our target audience’s needs.
Fabian de Kok-Mercado
Creative Director, Howard Hughes Medical Institute (HHMI)

Your clear, didactic visuals enabled us to open a vital dialogue with physicians, drawing their attention to a rare genetic disorder, its causes, diagnosis, and treatment.
Joel Bathe
Senior Product Manager,
Genzyme Canada
Genzyme Canada

The AXS team works hard to understand our needs and goals to best interpret them to create a product that consistently exceeds our expectations. I’ve hired AXS in the past and will always look for a way to hire them in the future.
Adam B. King
Global Branding and Marketing Communications Manager,
Heartware
Heartware

AXS Studio is one of the top multimedia companies in our industry. Partnering with them on a booth activity is different; they’re always thinking ahead and thinking proactively.
David Clark
Vice President,
D&J Electronics
D&J Electronics

The Ad Board meeting went very well and the feedback on the animated piece was outstanding, even from the biggest and most important critics—who said they had never seen anything that was so well done and presented the topic so perfectly! Thanks again for helping to make this vision a reality for us and for all of the effort that you put forth on this project.
Vicki Kelemen
Senior Director of Market Development, Amylin Pharmaceuticals
Who we've proudly worked with
Where our work has been featured
We support clients across all therapeutic areas. Science inspires us and we want to share it with others
Let’s work together
to tell your story
Why invest in Interactive medical and scientific experiences?
- Interactivity supercharges engagement and learning—actively involving users in your medical science content. As a result, they retain the information better than if they had only read or watched your content.
- To draw more attendees to your scientific conference booth. Well-designed, visually-striking interactive experiences are proven to increase traffic and engagement, compared with passive booth materials.
- To understand your customers better. With interactivity, HCPs press buttons, answer questions and make decisions. We record this information to provide you with valuable metrics about knowledge gaps and interest in your scientific content.
- To reach your audience through multiple channels. A single interactive experience can be deployed to your website, your congress booth, and MSL iPads for field use.
- It is a lasting investment. We design interactive experiences to be periodically updated for long lifecycles. For example, Build a Biosimilar was deployed to 34 scientific conferences and to MSL iPads over its 5-year lifespan.
The AXS Studio difference
What makes an interactive medical and scientific experience successful with healthcare providers and patients? At AXS Studio, we begin with a human-centered design approach that prioritizes your learning objectives and your audience. We incorporate elements of gameplay and discovery to spark curiosity, and finish with flawless execution and unparalleled customer support.

The right team
Delivering extraordinary medical and scientific interactive experiences requires an exceptional team: multidisciplinary, highly creative, and science-literate. Our in-house medical communication designers, animators and developers work hand-in-hand, fusing user experience (UX) design, science visualization, and programming to bring unique and inspiring experiences to life. With graduate training, certification, and 20+ years of experience communicating medical science, we ensure your content and visuals are accurate and inspiring from ideation to delivery.
Design for results
Healthcare agencies often start with a technology—AR or VR, for example—and design an interactive experience to fit. We believe it’s essential to start with your learning objectives, then design using the technology that best meets those objectives—and delivers the results you’re seeking. A great user experience is about feeling engaged with the content. At AXS, we use interactive technology—be it gesture tracking, augmented reality (AR) or holography—to facilitate that connection with your message.
Prepared for success
Getting a medical or scientific interactive experience through PRC or MLR reviews comes with specific challenges. For example, few medical reviewers have experience with gamified scientific education. We make it easy for them with expertly referenced review materials that are extremely clear and walk through each step of the user journey. Our team has shepherded countless “out-of-the-box” interactive experiences successfully through stringent review procedures.
Unexpected ideas
Interactivity connects people with your content in ways not possible with traditional media. We can present pathophysiology on a real-time scan of the learner, for example, or encourage them to seek out sites of disease within a virtual patient. With interactivity, multiple users can explore scientific content simultaneously and at their own pace. And physicians can simulate using a new medical technology rather than passively watching a video. At AXS Studio, we use design thinking to tap into our collective creativity and discover unexpected ideas for your next interactive experience.
Frequently Asked Questions
Simply contact us with your project information, including the timeline, budget and learning objectives. We’ll schedule a consultation meeting to review your goals and then provide a custom detailed proposal for your review.
Most medcomm and healthcare agencies outsource their client’s interactive projects to generalist developers. At AXS Studio, all design and development is done inhouse by our specialized team. Our medical communications designers, medical animators and certified medical illustrators all hail from scientific backgrounds. Each holds an M.Sc. degree in biomedical communications from one of the five CAAHEP-accredited graduate programs. These programs train digital artists in cell biology, anatomy, physiology, pathology and molecular visualization.
AXS Studio has 20+ years experience visualizing complex medical science for discerning HCP audiences. Our five Board-Certified Medical Illustrators (CMIs) ensure scientific accuracy in every interactive experience we create. We speak the language of science and are able to communicate complex concepts more accurately and clearly than other agencies and interactive companies.
The functional requirements and complexity of an activity are the biggest factors in cost. We account for your project budget when proposing a solution. Our fees include:
- Content writing
- Experience design
- Visual asset creation
- 2D and 3D animation
- Programming (web or game)
- Custom electronics
- Testing, deployment and support (on-site or remote)
Content creation, design and development are all handled in house by our long-standing team. This allows us to tightly manage all elements of production, while ensuring strict quality control and confidentiality. If you’re interested in a quote for an interactive experience or app, please get in touch.
Our quotes include ideation, content research and writing, UX and UI design, wireframing, 3D modeling and animation, programming, audio (voice, music, sound fx), application testing, support and training. During development, we prepare review materials for PRC or MLR and will handle Veeva (or other) submissions, if desired.
At AXS Studio, we’re committed to ensuring your science is communicated accurately. Our project managers, designers and artists immerse themselves in the literature supporting your content. We compile a comprehensive research deck to support the creation of all the visuals featured in your interactive experience. This guides our artists and ensures every visual in your interactive experience is backed by peer-reviewed research.
When clients first work with us, they often express relief after switching from their last agency. They cite previous challenges faced, including:
- Vendors who struggled to understand the project purpose
- Last-minute deliveries that cause undo stress
- Surprise technical problems at their conference booth
- Lack of support from their agency whenever something went wrong
- Unexpected charges to fix the agency’s own errors
In contrast, clients describe AXS Studio as “a talented and reliable partner”. They cite “the commitment [we] have to pulling off the impossible,” and the incredible quality and attention to detail in [our] work.” Clients express relief at no longer worrying about hitting deadlines, or surprise overages. And we collaborate well with booth and AV suppliers, who appreciate that we’re “always thinking ahead and thinking proactively.”
Biotech, pharma and medical device companies know they can rely on AXS Studio to deliver one-of-a-kind interactive experiences “that boost traffic and HCP engagement at … congress booths.” But don’t take our word for it, see what our clients say, themselves.
In general, development can take 3–6 months, depending on the scope. If you’re targeting a specific launch date, it’s important to start early—especially if your company has an involved MLR or PRC process. We suggest contacting us 6 months ahead of the due date, to be safe. Our project proposals include detailed timelines with key milestones so your team will know exactly when to expect key deliverables.
We advise aiming for 5 minutes or less. HCPs are busy people and have limited time to experience your content. Being concise and impactful provides the best value for their time investment. It also forces us to be highly targeted with key messages. We recommend prioritizing three key takeaways and an enjoyable experience for your audience.
- To begin, identify your primary audience and learning objectives. The audience will inform the complexity and language of your content. Eg., are you speaking to general practitioners or research scientists?
- Send us your key content resources. These may include scientific posters, training decks and journal papers.
- Our team will hold ideation sessions and present you with a proposal, including concept art. The concept and experience design may go through a couple of iterations at this stage.
- Once you approve the overall concept, we’ll prepare a content outline for your review and approval.
- Next, we’ll create a wireframe that shows the user experience, content (written and spoken) and visuals together. This document provides a clear picture of how the experience will look and function. It will help you get alignment from all stakeholders before development begins. After a round of review by your team, we’ll make any revisions and submit an updated wireframe for MLR/PRC review.
- Once the wireframe is approved, we develop alpha, beta and final versions of the functioning experiences. The beta version is typically submitted to MLR/PRC review, with any changes being pulled through in the final deliverable.
- Throughout development, we conduct system testing with your booth and/or AV vendor(s) to eliminate any chance of errors at launch time.
- Following MLR/PRC approval, we work with you to deploy the final interactive experience—whether to your congress booth, mobile devices, website or other medium.
Medical and scientific communications managers sponsor interactive experiences to educate visitors at their medical affairs conference booths. The content is often MOD and/or MOA education. Experiences designed for conference booths are often repurposed on medical affairs websites, providing ongoing support to HCPs.
Product and marketing managers commission interactive experiences to highlight new medical devices and technologies at trade show booths.
Sales representatives and Medical science liaisons (MSLs) use interactive medical or scientific apps in the field during physician meetings. These apps can be deployed to laptops, iPads and/or Android tablets.
Institutions including science centers and museums commission interactive scientific games and experiences to engage visitors in feature exhibits.
We’ll design your experience to function flawlessly without support by making it simple to use and providing on-screen and audio instructions. Nevertheless, an in-person facilitator usually increases participation and enhances the experience of your visitors. Many companies use trained facilitators for this reason. We can recommend a vendor that we’ve had great experiences with.
We typically handle the initial design of kiosks if they’re integral to the interactive experience. We then work with our clients’ booth vendors who build out the finished structures. Simultaneously, we coordinate with our clients’ AV vendors to spec optimal hardware and stress test the interactivity well ahead of the congress date. We ensure every aspect of your interactive experience is tested and production-ready, ensuring a smooth, stress-free launch for your team.
Want to learn more?
Privacy Preference Settings
To provide the best user experience, our website uses cookies and other related technologies (collectively referred to as "cookies") to store and/or access device information. Cookies are also placed by third parties we have engaged. Please see our Privacy Policy for more information.
Essential cookies Always active
These cookies are required for the website to function properly and enable core features such as page navigation, security, and saving your privacy preferences. Without these cookies, the website cannot operate correctly. You can set your browser to block these cookies, but some parts of the site may not function as intended. These cookies are necessary for the technical operation of the website and are exempt from consent under applicable data protection laws. They do not store personally identifiable information.
Preferences / Functional Cookies
These cookies enable the website to remember your preferences and choices, such as language, accessibility settings, and other personalization features, and to support optional website functionality such as embedded media and enhanced site features. They may be set by us or by third-party providers whose services have been added to our pages. If you do not allow these cookies, certain preferences may not be saved and some optional features or services may not function properly.
Statistics Cookies
These cookies help us understand how visitors interact with the website by collecting information like page visits, clicks, and loading times. We use this data to improve site performance and enhance the user experience. The information collected by statistics cookies is aggregated and therefore anonymous.
The technical storage or access that is used exclusively for anonymous statistical purposes. Without a subpoena, voluntary compliance on the part of your Internet Service Provider, or additional records from a third party, information stored or retrieved for this purpose alone cannot usually be used to identify you.
Marketing Cookies
These cookies are used to create user profiles to track users and display advertising on this or other websites. They work by identifying your browser and internet device and do not store directly personal information. Our website includes content from Blusky, Instagram, LinkedIn, and X (Formerly Twitter) to promote web pages (e.g. “like”, “pin” or “share”) on these social networks. This content is embedded with code from these networks and places cookies. This content might store and process certain information for personalized advertising. Please refer to the privacy statements of these social networks to learn what they do with your data. Blusky, Instagram, LinkedIn, and X (Formerly Twitter) are located in the United States.